 Foundations for Developing NEST Mark
Foundations for Developing NEST Mark
In September 2016, the FDA awarded a U01 cooperative agreement to the Medical Device Innovation Consortium (MDIC), a public-private partnership convening medical device manufacturers, researchers, regulators, payers, patients, academia, and health care providers to elevate regulatory science, to establish the National Evaluation System for health Technology Coordinating Center (NEST). NEST’s mission is to advance the sustainable generation and use of real-world evidence (RWE) for medical devices across their total product life cycle. NEST’s strategic direction and operations are informed by a range of stakeholders, including patients, clinicians, manufacturers, regulators, health systems, and payers.
Under the Medical Device User Fee Amendments of 2017 (MDUFA IV), NEST was tasked with foundational commitments to develop and implement a framework that supports the use ofRWE for medical devices. This involved establishing a governance structure, building the necessary infrastructure, and conducting pilot projects (Test Cases) to evaluate RWE’s potential in regulatory contexts. An independent third-party assessment further reinforced this initiative, identifying the strengths and limitations of RWE in informing pre-market review decisions for various device types (Timbie et al.). These foundational efforts set the stage for the transition to NEST Mark, a structured approach that advances the use of RWE in regulatory submissions.
Starting with MDUFA V in 2022, NEST shifted toward developing and implementing programs and tools that support regulatory submissions for medical devices with RWE. These programs draw from feasibility work completed during MDUFA IV and reflect input from NEST stakeholders.
How NEST Mark Advances RWD-Informed Regulatory Submissions
Building on insights from its feasibility work, NEST launched the development of NEST Mark—a process to structure the assessment and use of real-world data (RWD) for pre-market regulatory submissions for medical devices. This collaborative effort, involving stakeholders including the Center for Devices and Radiological Health (CDRH) and industry partners, is intended to develop clear, consistent methodologies to help ensure that RWD studies meet high standards of data relevance and reliability, is aligned with FDA Guidance Documents, NEST Frameworks, and other best practices.
The NEST Mark is intended to strengthen confidence in RWD’s relevance and reliability for meeting scientific and regulatory goals for medical devices, playing a key role in accelerating patient access to safe and effective health technologies.
NEST Mark: Real-World Data Source
High‑quality RWD is essential for generating relevant and reliable RWE to support medical device evaluation and regulatory decision‑making. To assist medical device stakeholders in assessing whether an RWD source (e.g., electronic health records [EHRs], claims, registries, or chargemaster databases) has the foundational characteristics needed for regulatory‑grade RWE, NEST developed the NEST Mark: Real‑World Data Source quality evaluation tool.
The NEST Mark: Real‑World Data Source approach is a structured, study‑agnostic evaluation of core data source capabilities, including accuracy, traceability, completeness, and related foundational attributes. This evaluation is intentionally independent of any specific research question, study design, or regulatory submission. Instead, it assesses the overall readiness of an RWD source to support RWE generation for medical device regulatory decision‑making.
Once a specific study question and curated dataset are defined, additional, study‑specific evaluations of data fitness and study design are required. However, when a dataset is derived from a source designated as a NEST Mark: Real‑World Data Source, users benefit from the prior completion of this foundational assessment, reducing redundancy and providing a transparent baseline upon which study‑specific work can be built.
The tool incorporates best practices, FDA guidance, insights from NEST Test and Implementation Cases (more details follow below), the NEST Data Quality Framework, the collective expertise of stakeholders across the medical device and RWE ecosystems, and piloting of early versions of the data quality tool with data sources, resulting in an expert-designed approach for evaluating RWD source quality.
Call to Action – Public Comment Period for NEST Mark: Real-World Data Source Quality Tool
NEST is currently seeking comments on the draft tool and proposed process from all stakeholders interested in data source quality for generating RWE for regulatory purposes. Respondents to NEST’s call for public comment should be aware of several factors when providing comments:
- NEST will continue to engage stakeholders, including the FDA, during and following the public comment period to ensure that the NEST Mark: Real‑World Data Source quality tool, assessment process, and rubric are aligned with stakeholder expectations.
- Accordingly, the draft tool issued for public comment is expected to evolve based on these engagements, as well as comments submitted by respondents to this open call.
- NEST anticipates that the final tool will be digitized to facilitate completion and review.
Please visit our open calls page to provide comments on the NEST Mark: Data Source Quality Tool during the public comment period, May 4th, 2026, to July 3rd, 2026.
NEST Mark: Future Work Using NEST Mark Implementation Cases
One of the major programs launched by NEST under MDUFA V is the NEST Mark Implementation Case program. Implementation Cases are designed to bring medical device companies, data sources, research partners, and FDA together to share the learnings across the life cycle of an RWE study to support a regulatory application for a medical device.
Launching Implementation Cases was crucial to inform the development of the NEST Mark: Real-World Data Source program. Current and future Implementation Cases will be used in combination with stakeholder engagement to determine if NEST should develop additional tools and/or programs under the NEST Mark umbrella once a study question has been identified. Figure 1 illustrates the next phase of the Implementation Case program.
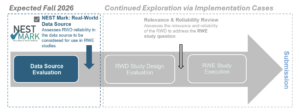
Contact us or review Open Calls to learn more about NEST Implementation Cases and how to engage with this pioneering work to improve regulatory submissions supported by RWE.
Citation:
Timbie JW, Kim AY, Baker L, Li R, W Concannon T. Lessons on the use of real-world data in medical device research: findings from the National Evaluation System for Health Technology Test-Cases. J Comp Eff Res. 2024 Sep;13(9):e240078. doi: 10.57264/cer-2024-0078. Epub 2024 Aug 16. PMID: 39150225; PMCID: PMC11367563. Lessons on the use of real-world data in medical device research: findings from the National Evalua…
